People
-

Principal Investigator, Ryan Roeder
Professor, Department of Aerospace and Mechanical Engineering
-

Prakash D. Nallathamby
Research Assistant Professor, Department of Aerospace and Mechanical Engineering
Doctoral Students
Tracie L. McGinnity (BioE)
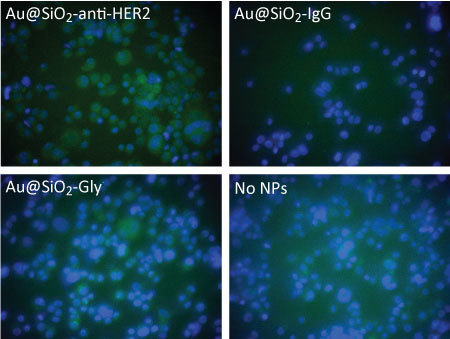
Molecular Imaging of Breast Cancer using Immunotargeted Radiographic Nanoparticle Imaging Probes, Hafnia Nanoparticles as a Novel X-ray Contrast Agent (NSF, Kelly Cares Foundation), 2013-present.
Project: Immunotargeting has recently emerged as a clinically successful therapeutic strategy for specific breast cancer subtypes. Gold nanoparticles conjugated with anti-HER2 antibodies have shown promise for radiographic imaging of HER2+ cells and tumors. Tracie is investigating the detection of limits for radiographic imaging of HER2+ breast cancer cells/tumors using gold-silica core-shell nanoparticles conjugate with anti-HER2. Hafnium oxide nanoparticles possess unique functional properties for use as an X-ray contrast agent, mid-infrared biosensor, and radiosensitizer. Tracie is investigating the synthesis of hafnia nanoparticles with tunable size, colloidal stability, and cytocompatibility for use as a radiographic contrast agent.
Awards: (1) Selected Participant, Clinical & Translational Research Course, NIH Clinical Center, 2017; (2) Professional Development Award, Downes Memorial Fund, University of Notre Dame Graduate School, 2017; (3) International Precious Metals Institute (IPMI) Student Award, 2017; (4) Student Travel Achievement Recognition (STAR) Award, 2017 Society for Biomaterials Annual Meeting, Minneapolis, MN; (5) 2016 Whitaker International Program Summer Grant, Whitaker Foundation, New York, NY; (6) Outstanding Graduate Student Teacher Award, Kaneb Center, Univ. of Notre Dame, 2016; (7) 3rd place, Ph.D. Poster Contest, 2nd Midwest Imaging and Microanalysis Workshop, Notre Dame, IN, 2015.
Felicia Roland (co-advisor, CHM)
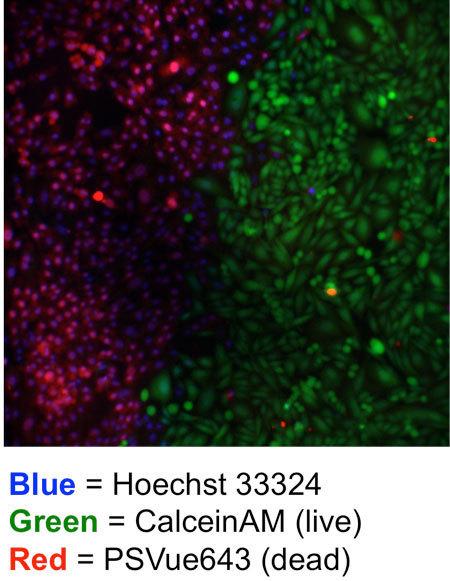
Theranostic Platforms for Imaging and Photothermal Therapy (Walther Cancer Foundation), 2016-present.
Project: Photothermal therapy (PTT) is an emerging new technique that is used for thermal ablation of cancerous tumors. PTT uses an exogenous chromophore to absorb laser light in the near-infrared spectral window to create a localized increase in temperature and resulting tumor cell death. Felicia is a part of the Smith group, which has discovered a new suite of organic photothermal agents, called croconaine dyes. These dyes are promising because they have a narrow excitation/emission peak at 800 nm to allow for tumor-specific heating. In addition, they have high photochemical stability and release energy primarily as heat. Felicia is collaborating with the Roeder Lab to develop Au-silica core-shell nanoparticles that consist of a gold core surrounded by silica, which can covalently encapsulate a bis(silane)-modified croconaine dye for PTT of cancer. Cells treated with these nanoparticles and irradiated with an 808-nm laser and ablated, as shown by a distinct boundary between cells that were in the direct path of the laser and those that were not.
Awards: Pre-doctoral Fellowship, Walther Cancer Foundation, Interdisciplinary Interface Training Project (IITP), 2016-2018.
Tyler E. Curtis (BioE)
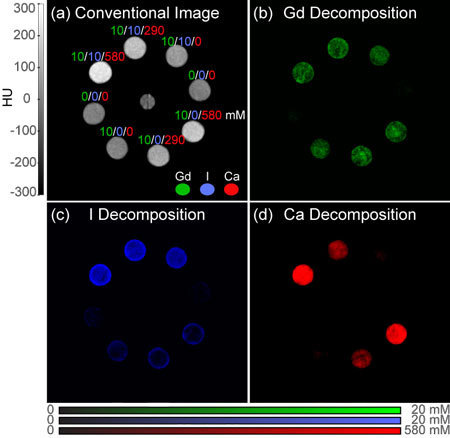
Quantitative Molecular Imaging of Mixed Compositions with Photon-Counting Spectral Computed Tomography (NSF, Kelly Cares Foundation), 2014-present.
Project: Photon-counting spectral CT has the potential to be a transformational new technology in clinical diagnostic imaging due to enabling an unprecedented capability of simultaneous quantitative molecular imaging and 3D anatomic imaging. Photon-counting detectors are able to measure energy-dependent differences in X-ray attenuation which can be unmixed for quantitative material decomposition. Decomposition of separate, non-overlapping materials has been demonstrated, but quantitative decomposition of mixed, overlapping compositions has not been previously realized. Tyler's research is focused on developing and evaluating methodologies for material decomposition, calibration, and image acquisition that enable quantitative molecular imaging of mixed compositions of multiple contrast agents and tissues with photon-counting spectral CT.
Awards: Outstanding Graduate Student Teacher Award, Kaneb Center, Univ. of Notre Dame, 2017.
Tyler A. Finamore (BioE)

Improvements in Breast Cancer Screening by Molecular Imaging, Non-Invasive Longitudinal Measurement of Collagen Scaffold Degradation Using Gold Nanoparticles (Kelly Cares Foundation, SJRMC, IPMI), 2016-present.
Project: Microcalcifications are the most common abnormality detected by mammography in breast cancer screening. However, the detection of microcalcifications and tumors may be masked by dense breast tissue. Bisphosphonate-functionalized gold nanoparticles (BP-Au NPs) were recently demonstrated to enable contrast-enhanced detection of microcalcifications in murine models; however, clinical translation requires further investigation using clinical imaging instrumentation in human-scale anatomic models. Therefore, the objective of Tyler's research is to develop an anatomic radiographic phantom mimicking varying levels breast tissue density for translational investigations of contrast-enhanced detection of breast microcalcifications using BP-Au NPs. The tissue model is designed to mimic a spectrum of tissue density, ranging from fatty (adipose) breast tissue to dense (fibroglandular) breast tissue.
Awards: International Precious Metals Institute (IPMI) Student Award, 2017.
Undergraduate Researchers
James Bathon (CBE)
Hydrothermal Synthesis of Nanoparticles, 2018-present.
Gino Domel (AME)
Prediction of Fracture Initiation Sites in Human Cortical Bone Using Nonlinear Micro-Finite Element Analysis, 2018-present.
Kyle Iwamoto (CBE)
Synthesis of Silica@Au Core-Shell Nanoparticles with Tunable Au Surface Density (DARPA), 2016-present.
Evan Gies (CBE)
Soft Hydrogels for Radiographic Phantoms Mimicking Varying Levels of Breast Tissue Density (Kelly Cares Foundation), 2017-present.